 The two reagents react with reducing functional groups in unique ways: Benedict’s reagent reacts with aldoses to form a red copper precipitate, while Tollen’s reagent reacts with aldehydes to form a silver, mirrorlike precipitate. Conversely, non-reducing sugars lack a free anomeric carbon.īenedict’s reagent and Tollen’s reagent are two reagents that are commonly used to detect the presence of reducing sugars. Reducing sugars can be identified through the presence of a free anomeric carbon, meaning it is not in a glycosidic bond and has a free hydroxyl group. 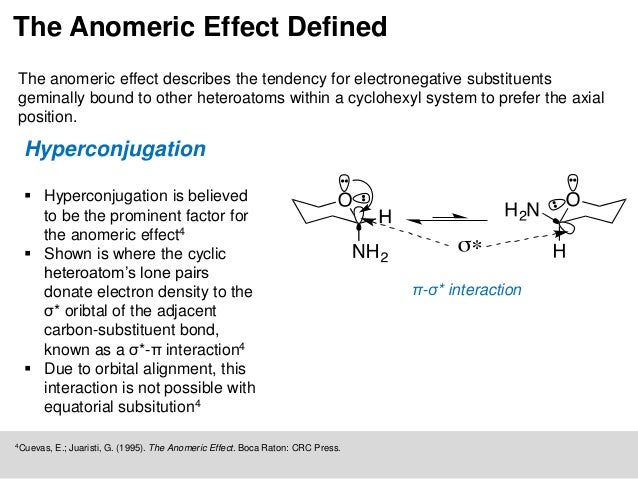 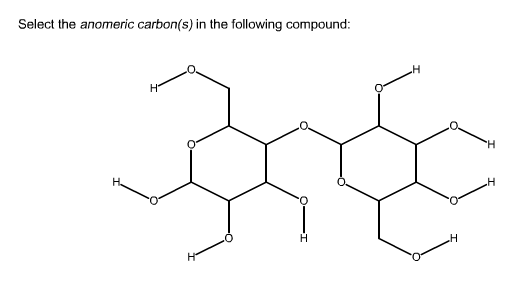
Sugars can also be described as being “non-reducing” or “reducing.” A reducing sugar is one that can act as a reducing agent. (The final ratios of each of these forms are usually affected by stereochemistry unique to each molecule.) This tendency to undergo spontaneous cyclization is known as mutarotation. As a result, many sugars can be found in various combinations of D-, L-, and linear forms. Note that as we convert the structure of the sugar from its two-dimensional to three-dimensional structure, the functional group that is pointing to the right of the Fischer projection will end up pointing downward in the Haworth projection.įor most sugars that are dissolved in a solution, this cyclization occurs spontaneously. A sugar is in its β-anomer form when the anomeric carbon’s substituent is on the same side of the plane as the highest numbered chiral center’s substituent. The anomeric effect is one of the most important factors influencing conformational behavior and also the reactivity of saturated heterocyclic systems. A sugar is in its α-anomer form when the anomeric carbon’s substituent is on the opposite side of the plane as the highest numbered chiral center’s substituent. 
In a cyclic sugar, the anomeric carbon is the carbon that has two bonds to oxygen.īe aware that a cyclic sugar can exist in two possible anomers: an ⍺-anomer and a β-anomer. While Fischer projections represent the straight-chain form of carbohydrates, you may also see sugars represented in their cyclic form as Haworth projections. If the hydroxyl group of the highest-numbered chiral carbon is on the left, it is in the L-configuration. If the hydroxyl group of the highest-numbered chiral carbon is on the right, the sugar is in the D-configuration. From that point of view, the stereoelectronic component of the anomeric effect plays a unique role in guiding reaction design.For sugars, the absolute configuration is designated using D- and L- nomenclature instead of the R and S system used in organic chemistry. This analysis paves the way for the broader discussion of the omnipresent importance of negative hyperconjugation in oxygen-containing functional groups. For example, negative hyperconjugation can unleash the “underutilized” stereoelectronic power of unshared electrons ( i.e., the lone pairs) to stabilize a developing positive charge at an anomeric carbon. In these situations, the role of orbital interactions increases to the extent where they can define reactivity. Even when many factors contribute to the observed structural and conformational trends, electron delocalization is a dominating force when the electronic demand is high ( i.e., bonds are breaking as molecules distort from their equilibrium geometries). Stereoelectronic thinking can reconcile quantum complexity with chemical intuition and build the conceptual bridge between structure and reactivity. Why are such questions inherently controversial? How to describe a complex quantum system using a model that is “as simple as possible, but no simpler”? What is a fair test for such a model? Perhaps, instead of asking “who is right and who is wrong?” one should ask “why do we disagree?”. We will use recent controversies regarding the origin of the anomeric effect to start a deeper discussion relevant to any electronic effect. We show that the complete hyperconjugative model remains superior in explaining the interplay between structure and reactivity. After providing the background related to the relevant types of hyperconjugation and a brief historic outline of the origins of the anomeric effect, we outline variations of its patterns and provide illustrative examples for the role of the anomeric effect in structure, stability, and spectroscopic properties. Herein, we will use a classic conformational “oddity”, the anomeric effect, to discuss the value of identifying the key contributors to reactivity that can guide chemical predictions. The task becomes even more difficult when multiple approaches based on different physical premises disagree in their analysis of a multicomponent molecular system. Understanding the interplay of multiple components (steric, electrostatic, stereoelectronic, dispersive, etc.) that define the overall energy, structure, and reactivity of organic molecules can be a daunting task. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
